 
It should also be noted that "percent" is simply the "fraction times 100", and so they are two different ways of stating the same relationship, and that it is a matter of convention to use percent for mass and fraction for the number of molecules, but you could also have "mass fraction" and "mole percent". Options for hiding the symbol or name of the elements provide a handy learning aid for memorizing the periodic table. 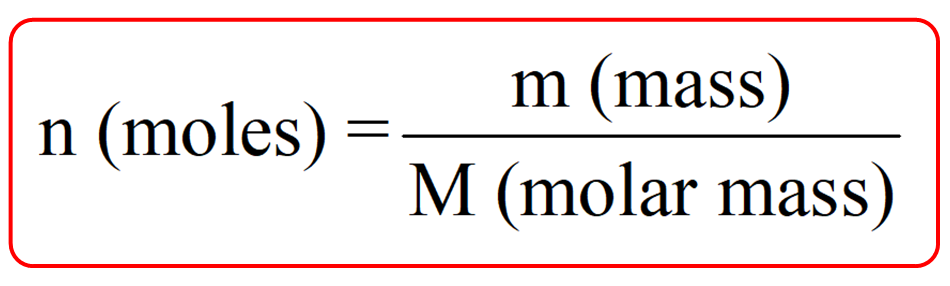 
The given mass of K (4.7 g) is a bit more than one-tenth the molar mass (39.10 g), so a reasonable ballpark estimate of the number of moles would be slightly greater than 0.1 mol. share my calculation Everyone who receives the link will be able to view this calculation. How do we determine the molar masses of molecules We do this by adding up the molar masses of all the atoms that comprise that molecule. For example, we used the concept of mole fraction to describe the partial pressure of a gas, where the partial pressure was the mole fraction times the total pressure, but that told you nothing about how close the molecules were to each other Use this periodic table for calculating molar mass for any chemical formula. Referring to the periodic table, the atomic mass of K is 39.10 amu, and so its molar mass is 39.10 g/mol. Molar masses of atoms are found directly on the periodic table. (b) 3.06 × 10 3 g of the amino acid glycine, C 2 H 5 NO 2. A 355-mL soft drink sample contains 0.133 mol of sucrose (table sugar). Determine the number of moles of compound and the number of moles of each type of atom in each of the following: (a) 25.0 g of propylene, C 3 H 6. Molarity is defined as the number of moles of solute in exactly 1 liter (1 L) of the solution: M molsolute Lsolution (1.4.3.4) (1.4.3.4) M m o l s o l u t e L s o l u t i o n. Note, the above can be used to describe the relative composition of homogenous mixtures (solutions), but don't really tell you about how close things are to each other. Molarity is defined as the number of moles of solute in exactly 1 liter (1 L) of the solution: M mol solute L solution M mol solute L solution. Molarity (M) is a useful concentration unit for many applications in chemistry. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
